What Is A Lone Pair In A Lewis Diagram
On other hand the lone pair explains the basicity of the ammonia molecule. A single shared pair of electrons is called a single bond.
Jump to navigation jump to search.
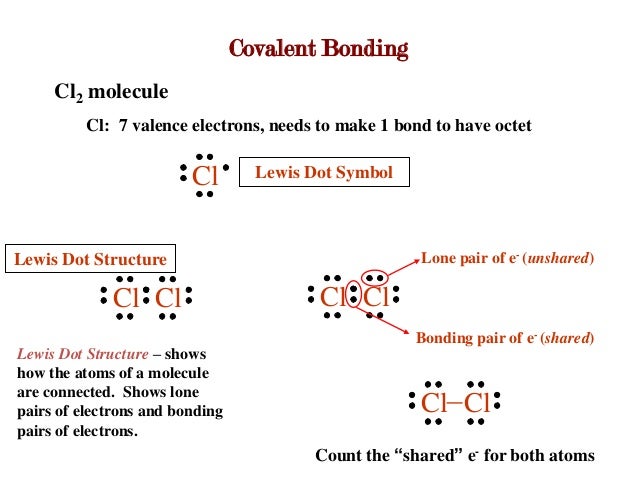
What is a lone pair in a lewis diagram. A lewis structure is a graphic representation of the electron distribution around atoms. Ammonium ion nh4 is a regular tetrahedron. In other words a lone pair is a non bonding pair.
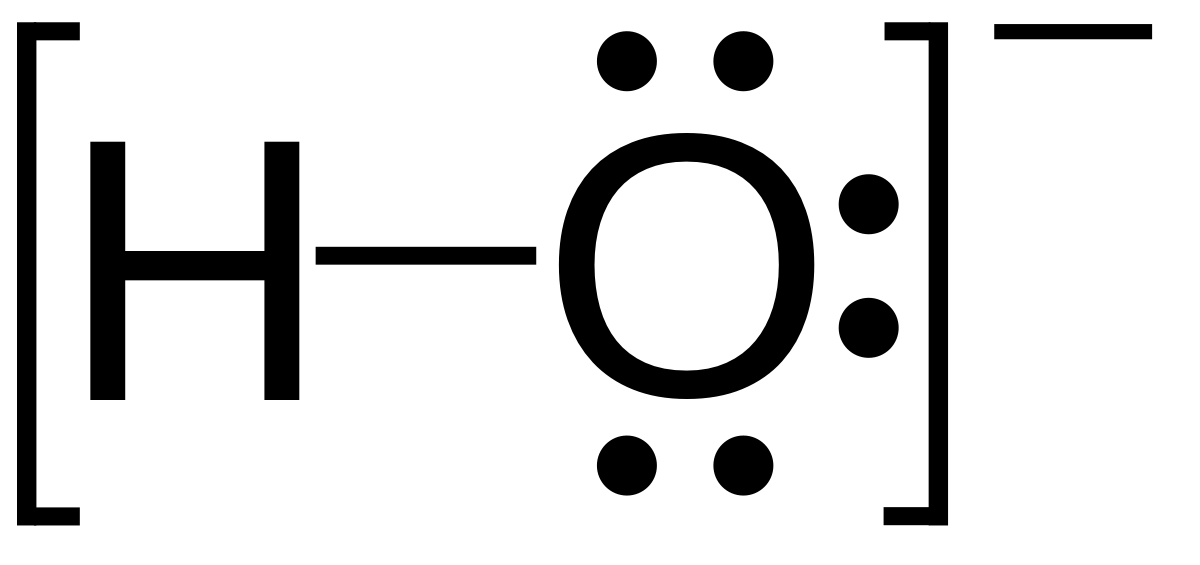
A dash or line is sometimes used to indicate a shared pair of electrons. The lewis structure indicates that each cl atom has three pairs of electrons that are not used in bonding called lone pairs and one shared pair of electrons written between the atoms. A lone pair consists of 2 electrons in the same orbital from the same atom which are not involved in bonding.
Lone pair is a concept used in valence shell electron pair repulsion theory vsepr theory which explains the shapes of molecules. Lewis structures also known as lewis dot diagrams lewis dot formulas lewis dot structures electron dot structures or lewis electron dot structures leds are diagrams that show the bonding between atoms of a molecule and the lone pairs of electrons that may exist in the molecule. There are lone pairs around the central atom so the geometry of seh2 is.
The total number of electron pairs both bonding pairs and lone pairs leads to what is called the electron domain geometry. There are lone pairs around the central atom so the geometry of pooh3 is. The lewis diagram for seh2 is.
The electron pair geometry around the n atom in nh3 is tetrahedral. On the other hand a bonding pair is 2 electrons from different atoms which overlap to form a pi or sigma bond. The lewis diagram for nh3 is.
The electron pair geometry around the se atom in seh2 is. When one or more of the bonding pairs of electrons is replaced with a lone pair the molecular geometry actual shape of the molecule is altered. However not all non bonding pairs of electrons are considered by chemists to be lone pairs.
And because the non bonding nitrogen lone pair lies fairly close to nitrogen it compresses the h n h bond down from 1095 at to approx. The reason for learning to draw lewis structures is to predict the number and type of bonds that may be formed around an atom. They are also referred to in the chemistry of lewis acids and bases.
 Lone Electron Pairs Introduction To Chemistry
Lone Electron Pairs Introduction To Chemistry
How To Recognizing Implicit Lone Pairs In Abbreviated Lewis Structures
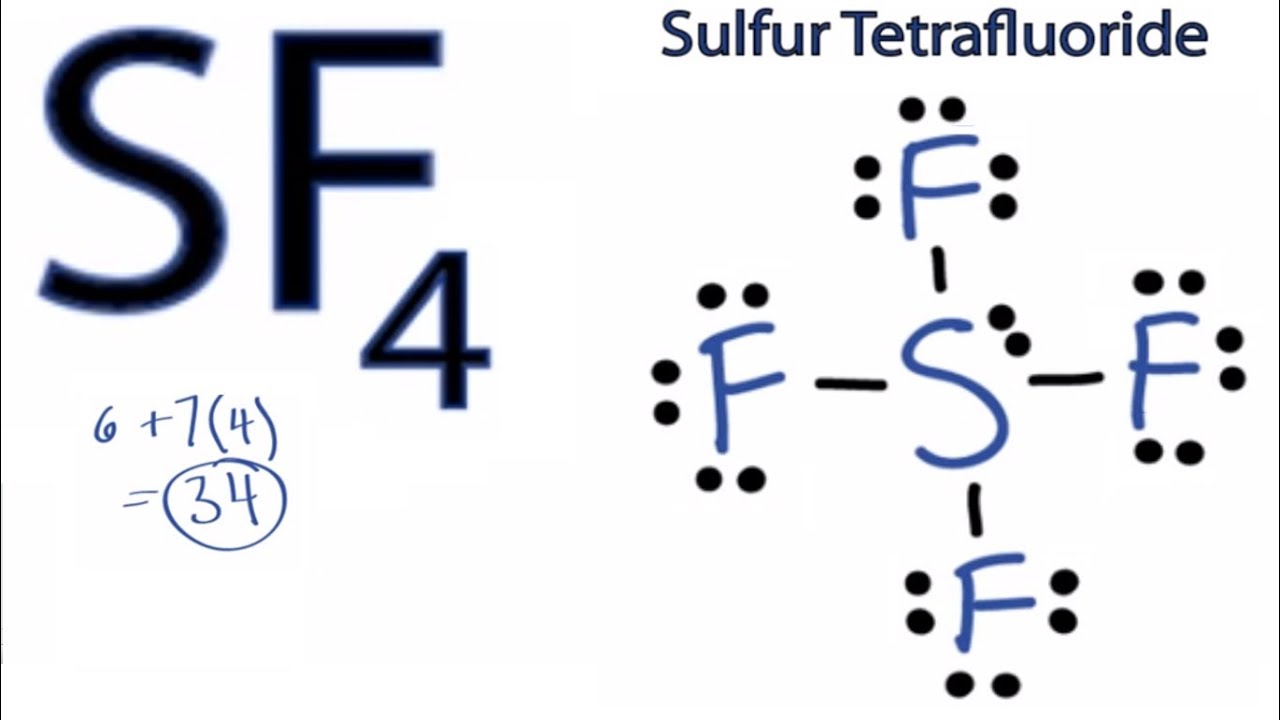 Sf4 Lewis Structure How To Draw The Lewis Structure For Sf4 Youtube
Sf4 Lewis Structure How To Draw The Lewis Structure For Sf4 Youtube
 Lewis Structure Molecular Geometry Atom Lone Pair Vsepr Theory
Lewis Structure Molecular Geometry Atom Lone Pair Vsepr Theory
 7 3 Lewis Symbols And Structures Chemistry
7 3 Lewis Symbols And Structures Chemistry
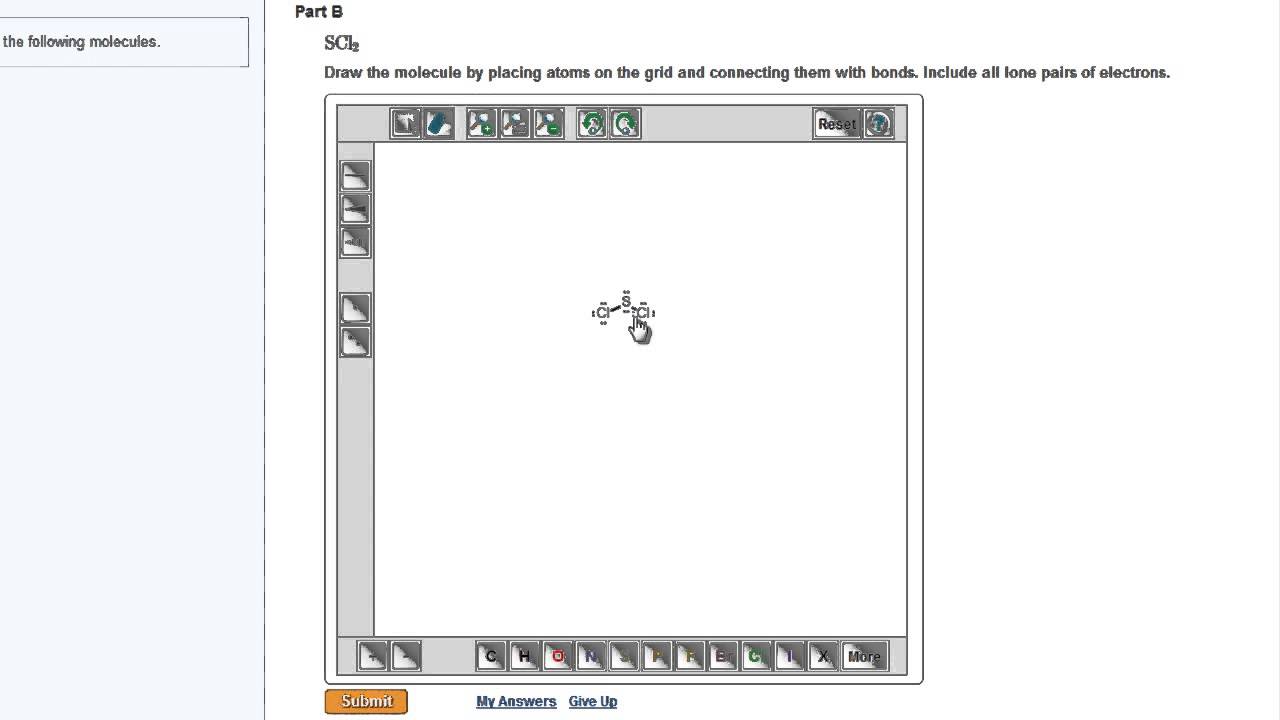 Masteringchemistry Drawing Lewis Structures Youtube
Masteringchemistry Drawing Lewis Structures Youtube
 What Is The Lewis Structure Include Lone Pairs Of Brf3 If Anyone
What Is The Lewis Structure Include Lone Pairs Of Brf3 If Anyone
Lewis Structure Of Brf5 Biochemhelp
 Brf5 Lewis Structure How To Draw The Lewis Dot Structure For Brf5
Brf5 Lewis Structure How To Draw The Lewis Dot Structure For Brf5
 Solved Pf3 Draw The Lewis Structure For Pf3 In The Box At
Solved Pf3 Draw The Lewis Structure For Pf3 In The Box At
Template Notebook Chemistry Labs C 10012 Molecular Shapes And Lewis
 Solution Add Lone Pairs To These Lewis S Chemistry
Solution Add Lone Pairs To These Lewis S Chemistry
 7 3 Lewis Symbols And Structures Chemistry
7 3 Lewis Symbols And Structures Chemistry
Lesson 9 Polarity Simple Cases
 Solved Draw The Lewis Structure Of The Following Molecule
Solved Draw The Lewis Structure Of The Following Molecule
 Solved Click The Draw Structure Button To Launch The Dr
Solved Click The Draw Structure Button To Launch The Dr
 Solutions To Chem 1 Exams 1996 97 2nd Midterm
Solutions To Chem 1 Exams 1996 97 2nd Midterm
 Solved Draw The Lewis Structure Of Clbr Showing All Lone
Solved Draw The Lewis Structure Of Clbr Showing All Lone

0 Response to "What Is A Lone Pair In A Lewis Diagram"
Post a Comment