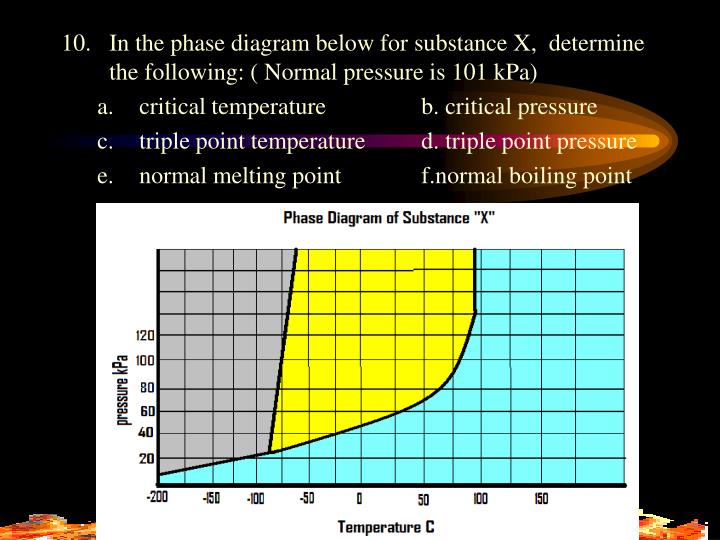
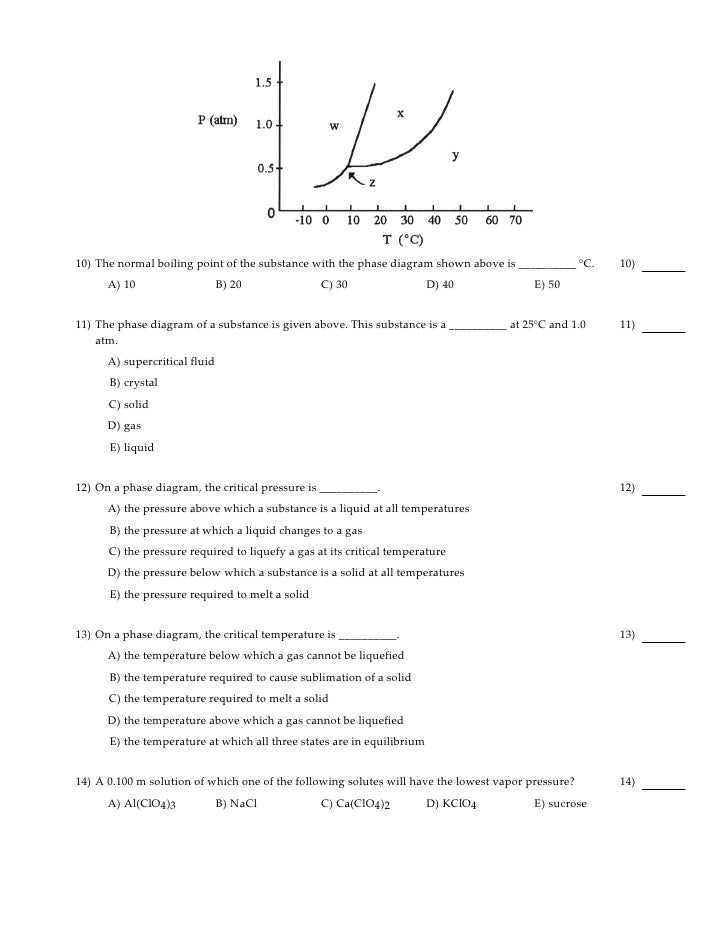
The Normal Boiling Point For The Substance In The Phase Diagram Below Is Approximately
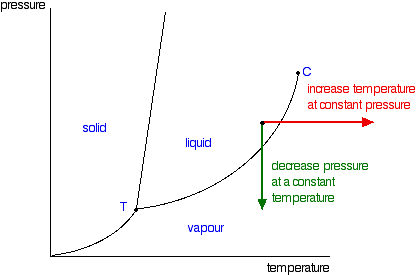
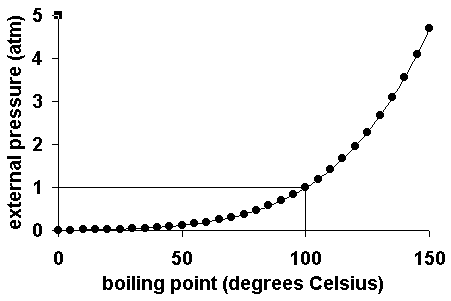
For example at 1 atm the boiling point is 100 c. The line from a to b is the vapor pressure curve of the liquid.
 Ppt Lesson 33 Energy And Phase Change Powerpoint Presentation Id
Ppt Lesson 33 Energy And Phase Change Powerpoint Presentation Id
Figure 1 a estimate the normal boiling point of the substance.

The normal boiling point for the substance in the phase diagram below is approximately. What is the triple point of this system. B estimate the normal freezing point of the substance. Entropy change on vaporization b.
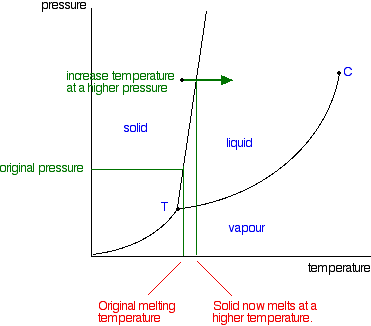
The point on this curve where the vapor pressure is 1 atm is the normal boiling point of the substance. Conditions necessary for sublimation c. When the pressure fell low enough the ice would then sublime to give water vapour.
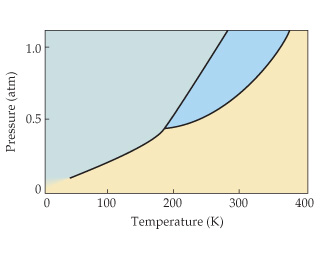
The figure below shows an example of a phase diagram which summarizes the effect of temperature and pressure on a substance in a closed containerevery point in this diagram represents a possible combination of temperature and pressure for the system. Approximately what is the normal boiling point of the substance. The approximate normal boiling point of this substance is 300 k imagine a reaction that results in a change in both volume and temperature as shown in the diagram below.
Refer to the figure and describe all the phase changes that would occur in each of the following cases. The phase diagram of a hypothetical substance is shown below. It represents the equilibrium between the liquid and gas phases.
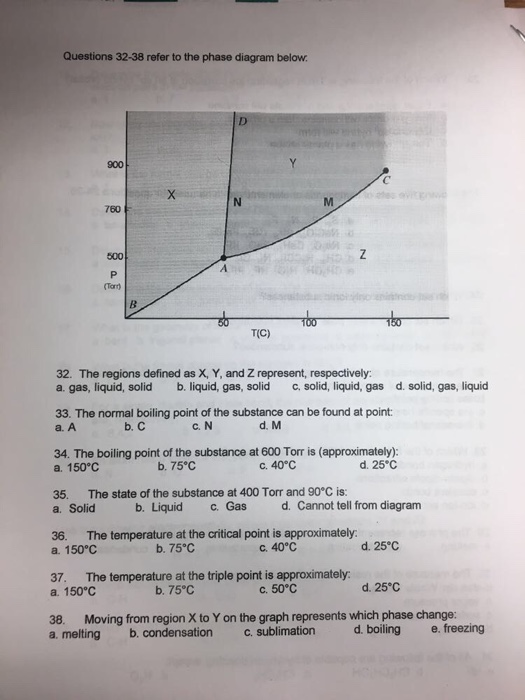
Express your answer using two significant figures. The normal boiling point of the substance represented by this phase diagram is approximately 2. Gas liquid and solid.
The phase diagram shows that the water would first freeze to form ice as it crossed into the solid area. Express your answer using two significant figures. This phase diagram provides sufficient information for determining the a.
Notice that the liquid vapor curve terminates at a temperature of 374 c and a pressure of 218 atm. Figure 1124 general shape for a phase diagram of a system exhibiting three phases. Part a water vapor originally at 0005 atm and 05c is slowly compressed at constant temperature until the final pressure is 20 atm.
This liquid vapor curve separates the liquid and gaseous regions of the phase diagram and provides the boiling point for water at any pressure. Imagine lowering the pressure on liquid water along the line in the diagram below. The phase diagram of a substance is shown below.
Express your answer using two significant figures. Normal melting and boiling points. Phase diagrams of pure substances.
Deviations from ideal gas behavior of the gas phase d.
 10 1 Intermolecular Forces Chemistry
10 1 Intermolecular Forces Chemistry
 Solved The Phase Diagram Of A Hypothetical Substance Is S
Solved The Phase Diagram Of A Hypothetical Substance Is S
How Are Vapor Pressure And Boiling Point Related Socratic
 Phase Diagrams Of Pure Substances
Phase Diagrams Of Pure Substances
 Phase Diagrams Of Pure Substances
Phase Diagrams Of Pure Substances
 Ap Chemistry Problem Set Chapter 10
Ap Chemistry Problem Set Chapter 10
 Boiling Point An Overview Sciencedirect Topics
Boiling Point An Overview Sciencedirect Topics
 Chapter 2a Pure Substances Phase Change Properties Updated 9 20 09
Chapter 2a Pure Substances Phase Change Properties Updated 9 20 09
 Answer The Following Questions Based On Th Clutch Prep
Answer The Following Questions Based On Th Clutch Prep




0 Response to "The Normal Boiling Point For The Substance In The Phase Diagram Below Is Approximately"
Post a Comment